Follow the instructions that came with your tool to adjust your readings. 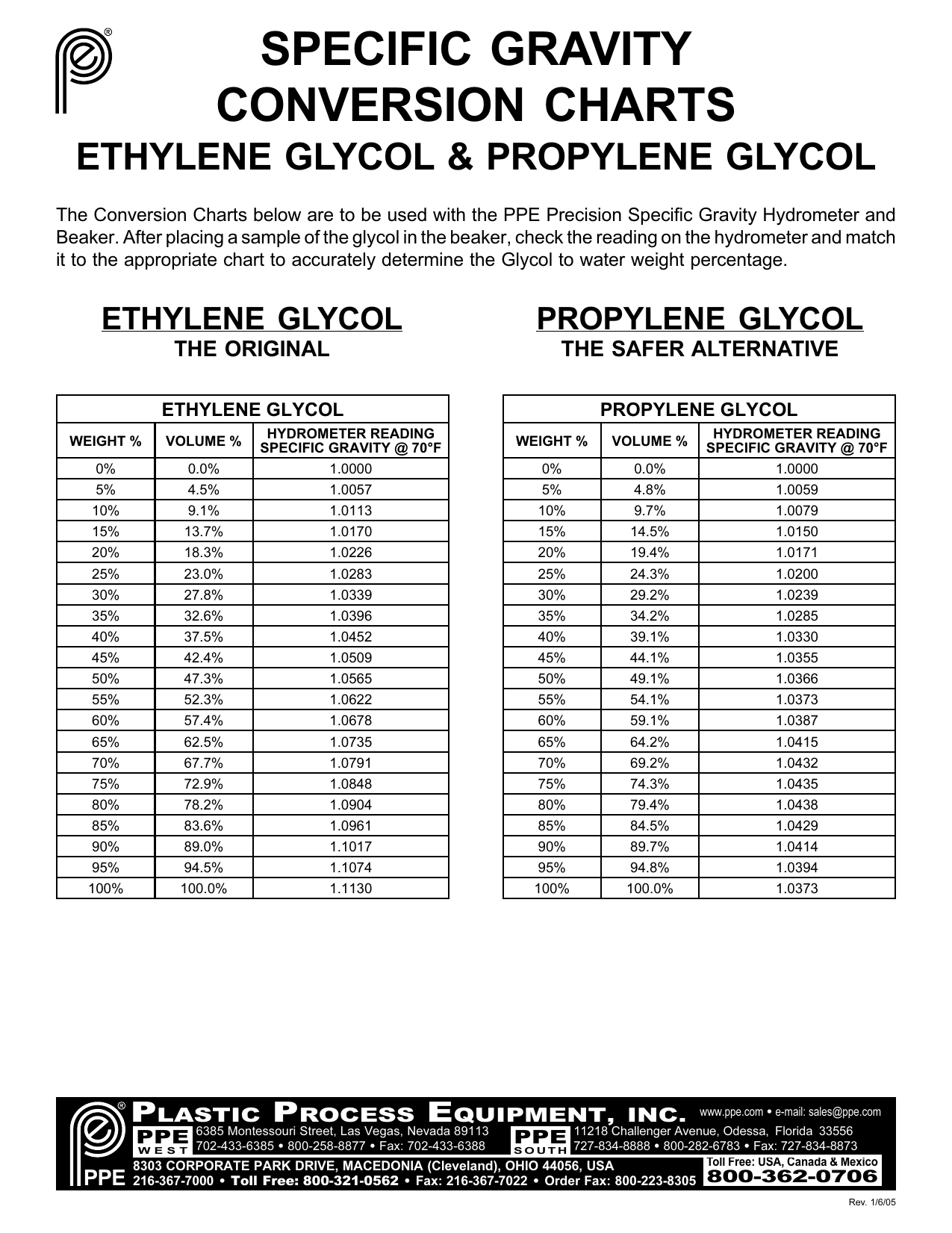
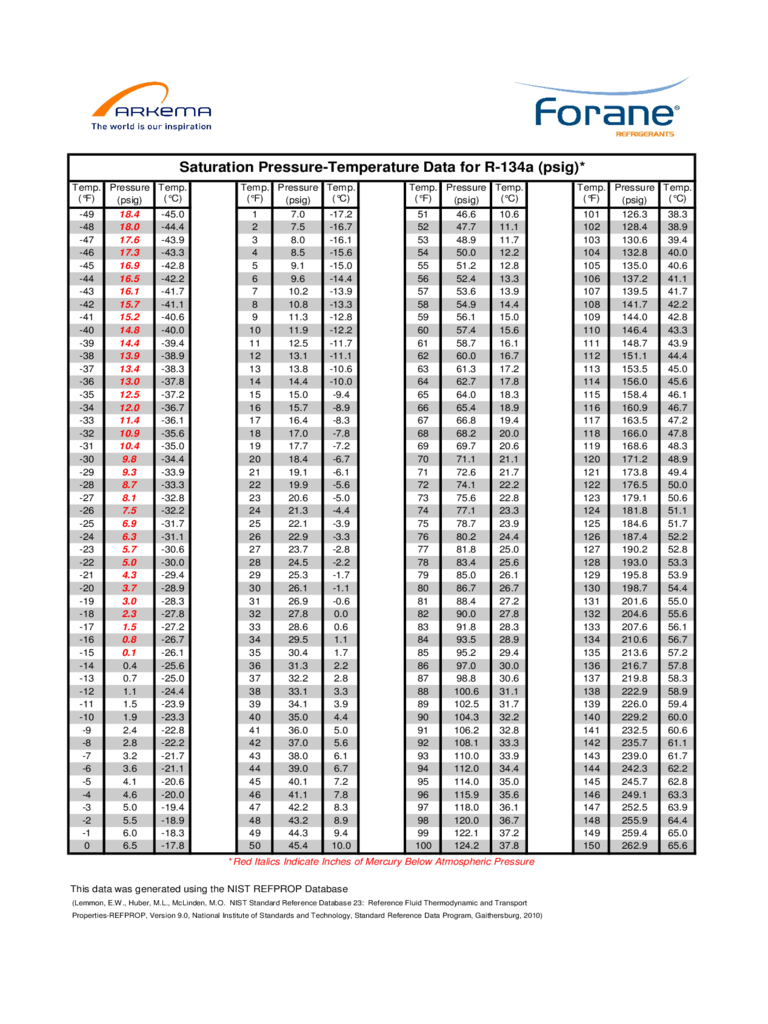
If the temperature on your thermometer is 65 F and your gravity reading is 1.210, adjust your reading to 1.206. For example, if the temperature on your thermometer is 93 F and your gravity reading is 1.210, adjust your reading to 1.214. Add or subtract four points (.004) to your reading for every 10 F (6 C) registered above or below 80 F (27 C). Use the conversion table on your tool to adjust your reading. Get the temperature from the thermometer.If you have a ball-or needle-type-hydrometer, follow the instructions on your tool's package for the reading. Jot down your reading on a piece of paper. To read the electrolyte specific gravity, read the number on the float's scale that intersects the surface level of the electrolyte.Maintain the hydrometer in a vertical position, and raise it to eye level.Make sure to pull enough of the liquid so the float in the hydrometer reaches around the center of the container and moves freely. Draw electrolyte into the hydrometer once more, starting with the first cell on either side of the battery.Repeat this four or five times to get the thermometer up to speed with the electrolyte temperature. Allow the liquid to sit inside the glass tube for a few seconds, and then release it back into the battery. 
Consult with a battery shop, if you need more information. If the electrolyte has a grayish tint, either a cell has degraded, or your battery has reached the end of its service life. You should be able to see through the colorless liquid. Release the bulb and let some electrolyte enter the hydrometer container.Remove the caps from the battery top, squeeze the hydrometer rubber bulb and submerge the tip into the electrolyte.Put on your acid-resistant gloves and goggles.RESOURCESĬlick here for a convenient specific gravity to salinity calculator ( data used to create this calculator is from LaMotte Company, Chestertown, MD, USA). Carefully place them into their protective packaging so they will not break. Once the reading has been made, clean the cylinder and hydrometer. The correct reading of this hydrometer is about 0.982. Just as reading the meniscus in a graduated cylinder, the user must take the reading where the plane of water is and not where the water clings up the neck of the hydrometer. Note that a meniscus forms on the neck of the hydrometer. Once the hydrometer has stopped bouncing up and down and the hydrometer is not touching the walls of the cylinder, a reading can be made. Assure that the hydrometer is not in contact with the sides of the cylinder and take the reading.Įxtreme care should be taken when reading the hydrometer it is very easy to misinterpret the scale.Note that the sample may overflow from the cylinder. Put the hydrometer with the bulb end down.Fill the glass cylinder with sample water.The user must be careful, though, to not break the the hydrometer or the cylinder as they are made from glass. The measurement made with the hydrometer can be used to calculate the salinity of a water sample. The hydrometers used for the Bayouside Classroom program range from 0.940 to 1.010 and are calibrated at 60✯/60✯. Hydrometers come with different scales depending on the characteristics of the liquid the user is measuring. Because specific gravity measures the mass of a liquid over the mass of distilled water, specific gravity is unitless. These hydrometers will be inscribed with “60✯/60✯” on their necks denoting that the reference density was measured at 60✯ and the sample should be at 60✯. Today, most hydrometers used in water quality testing are calibrated for with a reference temperature of 60✯. Classically, specific gravity was measured using the density of water at 4✬ (the temperature of maximum density for pure water). Because the density of a liquid changes with temperature, hydrometers are calibrated for different reference and sample temperatures. Specific gravity is the ratio of the mass of a liquid to the mass of an equal volume of pure water. A hydrometer is a tool scientists use to measure the specific gravity of liquids. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
